Identification of potential biomarkers for predicting the early onset of diabetic cardiomyopathy
Type 2 diabetes (T2D) is characterized by metabolic derangements that cause a shift in substrate preference, inducing cardiac interstitial fibrosis. Interstitial fibrosis plays a key role aggravating left ventricular diastolic dysfunction (LVDD) which has previously been associated with the asymptomatic onset of heart failure.
The latter is responsible for 80% of deaths among diabetic patients and has been termed diabetic cardiomyopathy (DCM). Through in silico prediction and subsequent detection in a leptin receptor deficient db/db mice model (db/db), we identified a biomarker profile that could potentially detect the early onset of DCM. Differential expression of Lysyl Oxidase Like 2 (LOXL2) and Mitochondria protein dehydrogenase (MPD), in both serum and heart tissue of 6-16-week-old db/db mice, confirmed cardiac dysfunction and correlated with a reduced LVDD as assessed by high-resolution Doppler echocardiography.
Principal component analysis of the combined biomarkers, LOXL2 and MTB, further displayed a significant difference between wild type and db/db mice from as early as 9 weeks of age. Knockdown experiments, utilising siRNA of either LOXL2 or MTB, revealed a decrease in the expression of Collagen Type I Alpha1 (COL1A1), a marker that contributes to enhanced myocardial fibrosis. Additionally, receiver-operating curve (ROC) analysis of the proposed diagnostic profile showed that the combination of LOXL2 and MTB resulted in an area under the curve (AUC) of 0.813, with a cutoff point of 0.824, suggesting the favorable positive predictive power of the model and further supporting the use of LOXL2 and MTB as possible early predictive DCM biomarkers.
In conclusion, we propose that the identified genes might be able to detect DCM asymptomatically and a follow up study is required using human serum with established DCM to confirm such findings.
Actualidad
Actualidad
- 25 de abril de 2024
El Hospital Universitari Dexeus inaugura el nuevo Instituto del Corazón Quirónsalud Dexeus en su compromiso...
El Instituto del Corazón Quirónsalud Dexeus reúne, en una misma planta de 650 m2, equipamientos renovados para el control de afecciones cardíacas complejas mediante los abord...Hospital Universitari DexeusCardiología - 25 de abril de 2024
La Fundación Jiménez Díaz ofrece consejos para controlar los síntomas del acúfeno y educar al cerebro...
Hoy se celebra el Día Mundial de las personas con AcúfenosHospital Universitario Fundación Jiménez Díaz - 25 de abril de 2024
El Materno-Infantil Quirónsalud Sevilla incorpora el tratamiento láser para la regeneración y la mejora...
Trata patologías como el dolor durante las relaciones sexuales, la sequedad, la atrofia o la incontinencia urinaria, así como lo relacionado con la estética genitalHospital Materno-Infantil Quirónsalud SevillaObstetricia y Ginecología - 25 de abril de 2024
El Hospital Quirónsalud Córdoba promueve hábitos de vida saludables entre los adolescentes
Profesionales del centro participan en el programa Stay Healthy de la Fundación Quirónsalud, a través de talleres sobre sueño, salud mental, adicciones a sustancias, nutrició...Hospital Quirónsalud Córdoba - 24 de abril de 2024
La Fundación Jiménez Díaz aborda el binomio pantallas y salud y analiza los riesgos de su uso inadecuado
El hospital madrileño celebra la tercera sesión de su ciclo de debates en Responsabilidad Social Corporativa “Salud, Personas y Sociedad. FJD Talks”Hospital Universitario Fundación Jiménez Díaz - 24 de abril de 2024
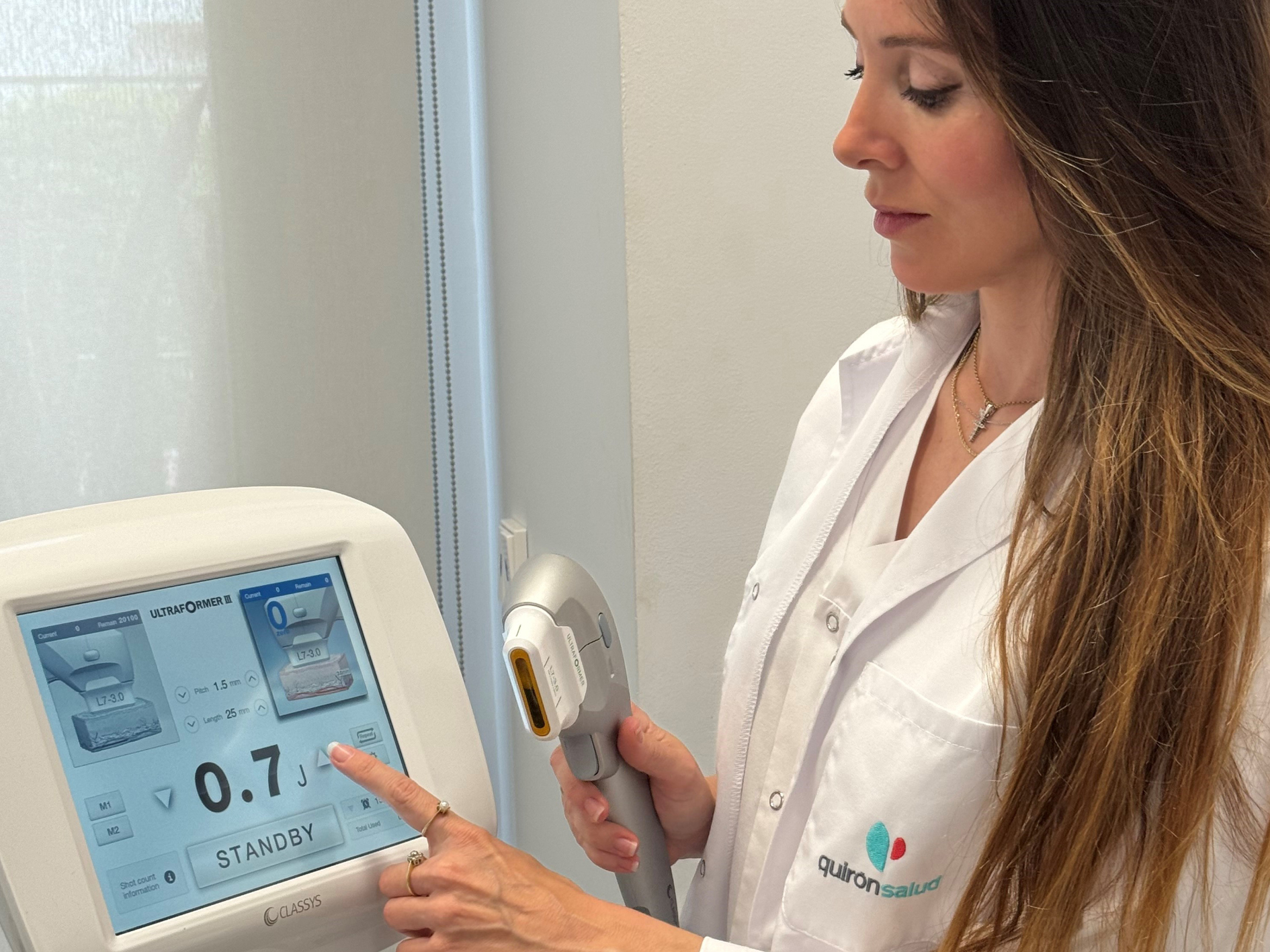
Ultrasonido de alta intensidad, el innovador tratamiento sin cirugía de rejuvenecimiento facial que no...
• Basado en ultrasonidos, no deja signos visibles tras el tratamiento por lo que permite hacer vida normal horas después de la sesión y se puede aplicar en los meses de veran...Hospital Quirónsalud Marbella - 24 de abril de 2024
Quirónsalud, Servicio Médico Oficial del Premier Padel Andalucía Sevilla P2
El torneo, que se disputa del 28 de abril al 5 de mayo, reúne a las principales figuras de este deporte, a nivel nacional e internacional.Quirónsalud - 24 de abril de 2024
El Hospital Quirónsalud Toledo celebra el Día del Libro con la lectura de ‘Las aventuras de Kenko’
Evadir del ámbito hospitalario, hacer disfrutar con la lectura y fomentar los hábitos saludables, los objetivos de la iniciativa.Hospital Quirónsalud ToledoPediatría y sus Áreas Específicas - 24 de abril de 2024
El Centro Médico Quirónsalud Valdebebas ofrece la posibilidad de conocer la edad metabólica
· Durante los viernes 26 de abril y 10 de mayo en horario de 9.00 a 14.00 horas y los miércoles 8 y 22 de mayo en horario de 15.00 a 20.00 horas. · Las personas interesadas q...Centro Médico Quirónsalud ValdebebasEndocrinología y Nutrición - 23 de abril de 2024
Quirónsalud Infanta Luisa celebra el Día del Libro con sus pacientes hospitalizados para promover los...
Quirónsalud Infanta Luisa regala libros a sus pacientes hospitalizados buscando amenizar así su estancia hospitalariaHospital Quirónsalud Infanta LuisaLogopedia - 24 de abril de 2024
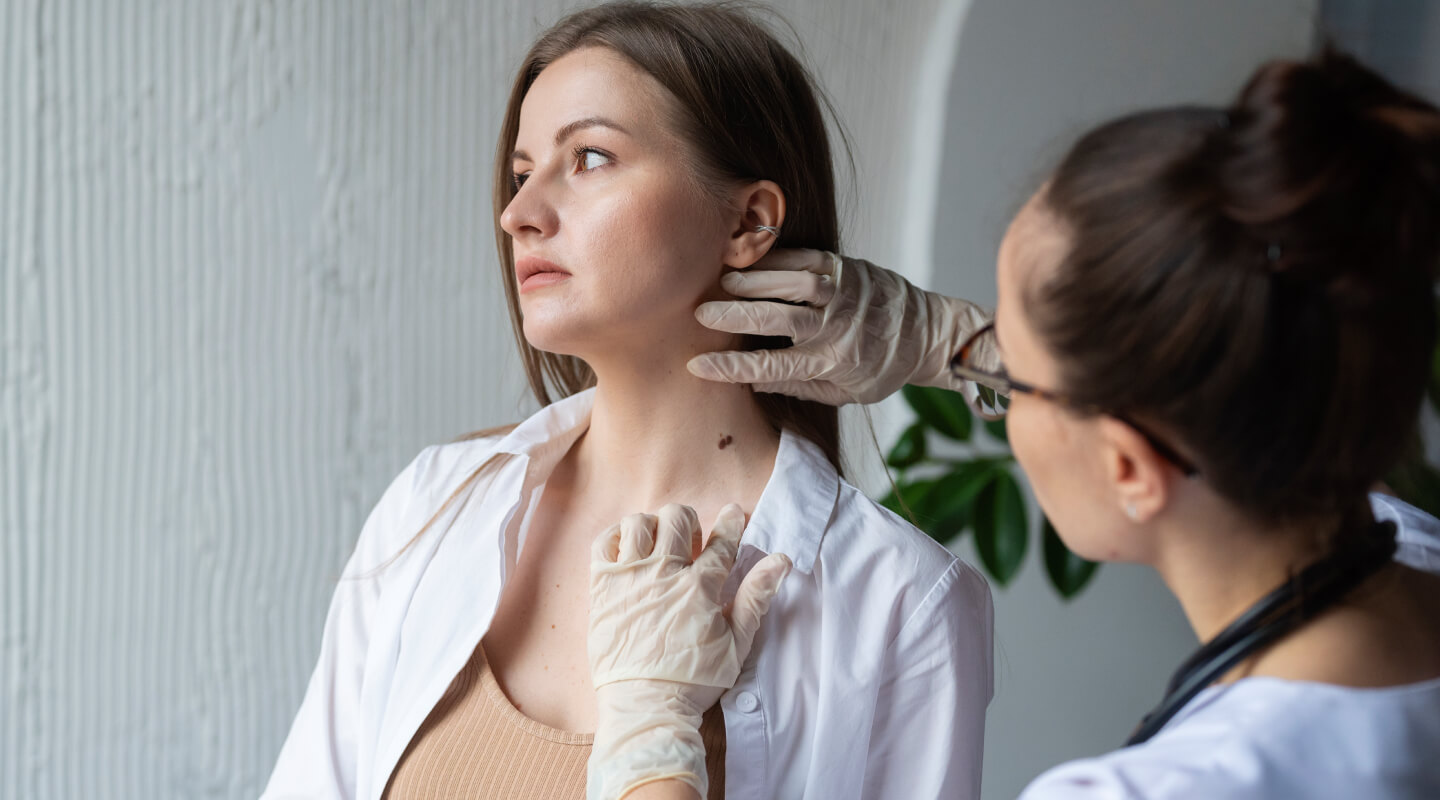
El Hospital Quirónsalud Badalona incorpora un nuevo servicio de Teledermatología para acelerar el diagnóstico...
El nuevo servicio agiliza la atención y respuesta médica, en 48 horas, posibilitando una actuación rápida sobre las lesiones que presenten indicios de malignidad.Hospital Quirónsalud BadalonaDermatología Médico-Quirúrgica y Venereología - 23 de abril de 2024
La Fundación Jiménez Díaz obtiene la acreditación de calidad más reconocida en estudios genéticos, la...
La acreditación, otorgada por la ENAC, refleja la calidad y buenas prácticas del Departamento de Genética del hospitalHospital Universitario Fundación Jiménez Díaz - 22 de abril de 2024
La Fundación Jiménez Díaz obtiene la certificación de calidad más avanzada en sus servicios de UCI Quirúrgica,...
La acreditación es una norma internacional de gestión de la calidad de los cuidados críticosHospital Universitario Fundación Jiménez Díaz - 23 de abril de 2024
Quirónsalud Málaga celebra el Día del Libro con sus pacientes
El personal de Enfermería ha repartido ejemplares cortesía de la Fundación Quirónsalud.Hospital Quirónsalud Málaga - 23 de abril de 2024
Celebramos Sant Jordi con Parada de libros y reparto de rosas
Hoy 23 de abril celebramos Sant Jordi en Quirónsalud Barcelona con el tradicional reparto de rosas y por primera vez con el estreno de una parada de libros tanto para trabaja...Hospital Quirónsalud Barcelona